AP CALL INFORMATIONFOR RESIDENTS
Contents
- 1 Recent Updates
- 2 Pager Operation
- 3 Overview of Call Responsibilities
- 4 Weekday Junior Call Responsibilities
- 5 Weekend/Holiday Weekend Junior Call Responsibilities
- 6 General tasks for senior weekend call
- 7 Accessioning
- 8 Handling Specimens
- 9 Autopsy
- 10 Call Room Resources
- 11 Tissue for Research
- 12 Neuro Tissue for Research
- 13 Cytology After-Hours Policy
- 14 Swapping Call Days
Recent Updates
06/22/2023 Merged call information:
The AP Junior Call Information document has been deleted and its information has been merged with this document.
Last updated: 06/22/2023.
Pager Operation
- To transfer the pager, call the MGH Operator and ask for p23305 to be forwarded to your pager number.
- In-house, dial 0 on any hospital phone.
- Outside the hospital, call 617-726-2000.
- To change your paging status, go to the Partners Telephone Directory (ppd.partners.org) → “Go to my entry” → “Change Paging Status.”
Overview of Call Responsibilities
Before you take call, make sure you know which Senior resident you're on call with and how to get in touch with them.
Responsibilities
- Assisting the senior resident in intraoperative consultations and rush biopsies.
- Answering clinical questions on pager 23305.
Time
- Weekdays/nights: 6pm – 8am (next day).
- Required to be in hospital from 6pm to 8pm.
- After 8pm, the junior resident can complete duties from outside the hospital by covering the AP pager (23305).
- If the Frozen section senior working during the day (8am-6pm) is the same senior covering call that night, the junior on-call may be asked to help as backup between 5pm-6pm.
- Weekends/holiday weekends: 8am Saturday – 8am next business day.
Schedule
- Weekdays only (Monday – Friday) for PGY1
- Weekdays and weekends/holidays for PGY2/3
Weekday Junior Call Responsibilities
When carrying the “23305” pager, you are essentially the “first call” for the department. Besides getting paged to do frozens, you may be asked to provide preliminary diagnoses on cases if available, to help clinicians determine how best to transport specimens to the lab, or to provide information about how a body can be transported to MGH for autopsy. If any issues arise that you are unsure as to how to handle, page your senior resident.
- Each weeknight, there is one senior resident (PGY3/4) and one junior resident (PGY1/2/3) who take call for the department. The junior resident’s call/shift responsibility begins and ends as outlined above.
- If the senior resident is covering the frozen lab during the day, you may be asked to start call shift at 5pm, as the 5-6pm hour is often a busy time. You should receive this request at least two weekdays prior to your call day. If you are unable to start at 5pm on that day, you must inform the Chief Resident.
- Your primary responsibility will be to assist the senior resident in evaluating and processing specimens that come in for frozen section (i.e. evaluate the gross, select the section to freeze, cut and stain the slide, accession the case). In general, it is better to page the senior resident when a specimen comes in before cutting into it – they are less experienced at interpreting frozen sections than the attendings who cover the lab during the daytime, and often prefer to evaluate the specimens for themselves and help with selection of sections for frozen section processing.
- Transfer the pager at 6pm (with no end time). See the “Pager Operation” section for further details. Never leave the pager unattended!
- Before 8pm: the surgeons expect us to be available in-house until 8pm so that there is essentially no delay between the time they call and the time we are available to do a frozen.
- After 8pm: if any frozens come in, the surgeon will page the 23305 beeper and should be understanding when you tell them that we are not in-house and to anticipate a 30-40 minute delay until the frozen can be performed.
- Make sure the daytime Frozen section resident has picked up the pager at 8am the following morning.
Patient care transfer between the daytime frozen section resident and the on-call resident (i.e. at 6pm or possibly 5pm)
- Ask the frozen section resident about any ongoing cases. If the frozen section resident is still busy at 6pm, make every effort to relieve them right at 6pm (finish the frozen they are currently working on, accession cases that may have recently come in, put chucks into cassettes, etc).
- If it is exceedingly busy in the frozen lab at 6pm, the frozen section resident is strongly encouraged, though not obligated, to stay and help.
- The frozen section resident SHOULD NOT leave without making sure that all the specimens from the day have gross descriptions!
- If frozen sections on a chuck are still in the cryostat and have not been taken down, it is the FROZEN SECTION RESIDENT’S RESPONSIBILITY to make sure that they are clearly labeled with the patient’s name and frozen section number. If they are not clearly labeled, the frozen section resident is responsible for taking these frozens down, labeling them, and putting them with the correct case even if it is after 6pm.
- Do not leave frozen tissue in the cryostat overnight. The cryostat has a thaw cycle and the specimen will be damaged.
- Before leaving for the day and especially on Friday, the frozen section resident should ascertain that there are no questions about the specimens that were handled during the day from prosectors. This is particularly true of large or complicated specimens, specimens where several frozens were taken, and/or those where the gross description was sparse due to time constraints.
Checklist of things to do in the Frozen section lab before you leave for the night
- REMOVE the blade in the Cryostat.
- Turn off the Cryostat light and shut the cryostat lid/door. Cover alcohol jars.
- Cover all staining materials.
- Clean up blood and tissue from Frozen section benches.
- Make sure all used frozen section chucks with tissue in them have been removed from the Cryostat and put into a cassette (labeled with FX sticker) and are placed in a container with formalin OR saved frozen tissue should be placed in a labeled bag and put in the -80C fridge in the histology lab to go to Warren 5.
- Empty all cubbyholes above the staining bench and place specimens in the “open” cubbyholes above the cubbyholes where specimens to be grossed by residents are kept.
- After accessioning new cases, write them in the log book (place accession sticker on front page with # of frozens done) and place photocopy the requisition sheet in the folder with the frozen slides.
- Turn microscope and room lights off.
Weekend/Holiday Weekend Junior Call Responsibilities
Weekends
- On non-holiday weekends, one junior resident (PGY2/3) and one senior resident take call for the department. The junior resident’s responsibilities include assisting the senior resident with frozen sections, triaging/prepping of specimens, and covering the 23305 pager.
- Timing: Saturday at 8am – Monday at 8am
- The junior resident must be present in the hospital on Saturday from 8am until 2pm. After 2pm on Saturday, there is no need for either resident to be physically in the hospital except to perform required duties (frozens, triaging/prepping, loading processors) and call may be taken from home.
- The junior resident is responsible for triaging all specimens that come in on the weekend. The senior resident is available and should be called with any questions. The resident should look for specimens in the front refrigerator, the basket under the front window, the frozen lab cubbies, and generally around the lab area. These are some specimen handling guidelines:
- Any specimens that belong in core lab or microbiology should be carried there in person as soon as possible (these labs are located on Gray 5).
- Any rush or clinically urgent cases should be accessioned, dictated, and grossed. “’This includes Neuropath specimens (except temporal lobe excisions or other large excisions) and other small specimens that have a frozen section performed on the weekend.”’
- Any specimen which requires special studies (e.g. a POC requiring cytogenetics) should be accessioned, and material should be taken for special studies.
- Lymphoma workup, including frozen assessment, should be performed if necessary.
- Large specimens
- Breast specimens should not be grossed unless you are contacted and explicitly asked to gross a breast by the breast attending.
- Other specimens do not need to be grossed but enough preliminary work (proper specimen preparation and fixation) must be done so that the specimen can be effectively grossed on Monday.
- If it is a specimen that is routinely inked, it should be inked at least in areas that are going to be disrupted by opening the specimen and in areas where it may be difficult to reconstruct what was true margin after formalin fixation,
- Note and/or mark any external findings that may be diagnostically important but more difficult to see when the specimen has been opened and fixed (like small serosal nodules).
- Non-reproducible measurements should be taken.
- This doesn't mean all measurements - just the ones that matter and cannot be reliably performed after your specimen handling/formalin fixation.
- Examples: weight of uterus - yes, length of cervix - no; how far colon tumor is from margin if it is less than 5 cm - yes; if it is really far - no.
- The specimen should be opened, photographed if necessary, and placed in formalin (but not accessioned) and placed on the shelf under the grossing lists next to the scanner.
Weekend Junior Call Workflow/Check List
- Saturdays, must be in-house 8:00 AM – 2:00 PM. Sundays can come when you would like (no loading the processor until 2:00 PM).
- Move placentas from the drop-off window to the Blake 354 fridge bottom shelf. There is no need to gross placentas (NICU or otherwise) unless contacted and explicitly asked by Drucilla.
- Check individual biopsy reqs in the larger bags dropped off at the window and in the metal basket next to the copier for RUSH cases. If present, have the senior resident triage with the clinical team as usual.
- Don’t look at the bags by individual accessioners desks.
- Check the fridge for specimens to prep when you get in an throughout the day.
- If prepping instructions are not in the AP Call Information document, check specialty-specific grossing manuals on LearnPath.
- Breasts do not need to be grossed unless you are contacted and explicitly asked to by the breast attending. Mastectomies: section through and put in formalin; if per diem PA (often Donna) will be in, email them and kindly ask to gross.
- Sometimes there is a PA grossing on the weekend. They are there to help you and you can ask them questions. The calendar for the per diem PA is on the wall to the right of Jenn’s desk.
- Don’t forget to put formalin stickers on your specimens!
- Leave prepped specimens on the table underneath the grossing lists next to the scanner.
Loading the Processors
- The tissue processors run on Saturday and Sunday on a regular weekend. The first time you take call on a weekend, ask the senior resident to help you with loading the processors. If processors are loaded before 5pm, there should be a histotechnologist in the lab who can help you with questions. When loading the processors on weekends, it is necessary to document the cassettes submitted to histology by taking a picture of the slide basket and printing it. This may be done using the specimen camera on Blake 3. Leave the printed picture on top of the processor you have loaded, labeled with date, time, and your initials. You do NOT need to scan the cassettes to the processor.
- Processor #1 (the one behind the door) should not be used as it is the processor for neuro/CJD cases.
- When loading the processors, make sure to pick a processor that is not currently being cleaned, or has low fluids (which will be designated on the main screen).*Specimens should only be run on 4-hour (small specimens) and 8-hour (large specimens) programs on the weekend. Select either run immediately or set to finish by 3:00am the next day (usually run immediately is the preferred method). If there is a histotech, you can ask them what the best end time is.
- Small biopsy specimens should not go on the 8-hour run as the tissue will become brittle and “overcooked.”
- The histology lab is staffed 7:30 am-4:00 pm on the weekends, and there is often someone coming in at 3:00 am on Sundays, so it is generally safe to set the tissue processor for 7:30 am, but if there is something urgent Saturday night, email William and let him know and he will let you know if there’s 3:00 am staffing on Sunday.
- Histology staff will start at 3:00 am on weekdays.
- If you leave cassettes in formalin over the weekend (I.e. if you had to gross a breast or something) you can bring the container into the histo processor room with a note of when the tissue went into formalin, and it will be loaded when ready.
- Cassettes in decal are checked by histology on Sunday, so you do not need to check them.*Inquiries about weekend rush cases should be directed to the senior resident on-call.
- Low threshold to call the contact numbers posted in the histo lab, including William Anim (617-962-2793), who is very helpful.
Holiday Weekends
- As on non-holiday weekends, timing is from 8 am the first day (typically Saturday) until 8am the next business day.
- On holiday weekends (hospital observed, 3-day weekends), the call responsibilities are divided between two junior (PGY2/3) residents. One senior resident is on call for the entire holiday weekend.
- Call responsibilities include the typical weekend duties (see above).
- The two junior residents may decide between themselves how to share the call, as long as the pager is always covered.
- On holiday weekends, usually the processors run once during the weekend and then the evening before the next business day. Check with the histology lab on Friday when the processors will run over the weekend before making your plans to load. In general, the processors should be loaded once before the first run, and then the day before the next business day (so if the holiday day is Monday, on Saturday and Monday, with Sunday being optional). Check the decal rack before loading.
General tasks for senior weekend call
- Triage rushes (Friday) – make a list of the cases that will be Saturday rushes so you can keep track (I’ve attached a template for this you can use)
- You will receive emails starting around 10am on Friday with Rush cases.
- The accessioners will title the email as Monday, Same day, or Triage rush.
- For Triage rush cases, when you receive the email, send an email (or page) to the ordering clinician to ask if it is a Saturday or Monday rush. For example…
- Hi Dr. ___, We received a rush pathology specimen for pt: ____, MRN: ____. Is this a Rush for Saturday, or for Monday morning? Thank you, _____
- Reply all to the original email to say if it is a Saturday or Monday Rush, this will alert the attending if they need to come in on Saturday. If it is a Monday Rush, tell the accessioner to re-accession the case to the Monday team (by default, all weekend rushes will be accessioned to you). Double check the accessioning of all the rush cases, however, because if the accessioning doesn’t change, it will go to your mailbox on Monday and will delay the case!
- Go up to histology in the morning and remind them what rushes you need that day. Gather rushes from histology around 10-12am (Sat) and deliver to attending mailboxes (double check your list to make sure you have gathered all appropriate Rushes). Let the attending know the case is ready. Sometimes they will want you to show the case virtually via Teams.
- Deal with any Frozens
- Make sure specimens are prepped/processors loaded Sat and Sun (by junior)
- Deal with any new rushes (have junior gross them, load on processor, and send email to Rush block/relevant attendings and residents). Specifically, if there is a request for a Sunday read, make sure this is approved by the weekend on-call attending since they will need to come in on Sunday and review. You will be responsible for coordinating with histo to ensure someone is here to take care of the Sunday rush. On Sunday, go up to histology in the morning, get the case, and deliver to the attending’s mailbox. Let the attending know the case is ready.
- To ensure you do not miss any rushes because you’ve been left off of the initial email, add the “MGH Accession Log by Specimen” to your favorites on Copath.
- MGH Accession Log by Specimen >> load criteria >> Friday or Sat Rushes (depending on day) >> Ok >> Ok
- ”’Important senior call reminders”’: The senior on call should ALWAYS page the neuropathology attending when you are alerted to a potential neuro frozen section after hours and over the weekend during call. Do not only send an email; the staff must be made aware immediately via a page.
Accessioning
How to accession a specimen
Frozen specimen quick accessioning
Bone marrow== Frozen Sections ==
Tips for Cutting
- Don’t over-freeze in liquid nitrogen. Typically 12-13 seconds in liquid nitrogen followed by slow freezing in the cryostat works well.
- Breast sentinel nodes are often fatty and thus challenging to cut. Here are some suggestions from our histotechs:
- Orient the specimen such that lymphoid tissue is near an edge of the chuck, rather than in the center with the fat near an edge. This way the blade cuts into lymphoid tissue first, rather than fat first.
- It’s easier to pick up a section when you have a little extra OCT edge to “pull” on.
- When you’re into the specimen, look and confirm that you’re cutting into lymphoid tissue first. If not, re-orient the chuck so that the blade cuts into lymphoid tissue first.
- Keep the blade cold. Use the gauze to drip liquid nitrogen onto the blade. Per the histotechs, this works better than freezing the chuck/specimen.
- If all else fails, try increasing the thickness of the section. The histotechs don’t usually go above 6-7 microns, but sometimes it takes us residents 8 microns to get something on the slide.
Unstained slides for molecular
- This applies to lung and neuro rapid molecular cases. The CID lab is expanding the catalogue of rapid molecular testing they are able to perform. They have requested additional slides so they have sufficient material to workup these extra tests. The workflow will not change significantly except for the pathologist should clearly indicate to the histotech how many slides to cut after reviewing the frozen slide. If there is sufficient material, please increase to 19 unstained + 1 H&E. If there is scant tissue, revert to 9 unstained + 1 H&E. It is okay to cut 19 unstained sections with two sections per slide.
- During weekday work hours, the histotech in the frozen section lab will cut the slides and then page Rapid Molecular (29059). After hours and on the weekend, the on-call team needs to cut the sections as outlined above. The unstained slides and the H&E recut (not the H&E used for frozen section diagnosis) should be placed with a copy of the requisition on the window shelf between the six-headed scope and the accessioning computer in the frozen section lab. Please keep the original paperwork with the specimen. An email must be sent to Nick Jessop at NJESSOP@PARTNERS.ORG indicating that there are slides for molecular pickup the following business day morning. In addition to the email, you can still page Rapid Molecular as they check the pager first thing Monday morning, but molecular staff are not available during the weekend.
- The slides do not require refrigeration. They can be left at room temperature until pickup the following business day morning.
Handling Specimens
Specimen Dropoff
- Surgical specimens: after hours/weekends, have surgeon/clinician drop it off in the front fridge and sign the book.
- Cytology: after hours/on the weekend, leave the specimen in the bin labeled 'cytology' in the front fridge.
RUSH Specimens
- Page/Question: Clinician wants to rush a specimen.
- Action: Call the histology lab (x4-1425) and provide them with the case number to request a priority change to make sure the case is processed as a Rush and email the RUSHblock email group <RUSHblock@partnershealthcare.onmicrosoft.com>.
- Note: The case WILL NOT be rushed if the histology lab is not notified. Do NOT change anything in CoPath.
- Page/Question: Clinician wants to rush a cytology specimen.
- Action: Email Ron Arpin <rarpin@mgh.harvard.edu>.
- Page/Question: Clinician wants read of rush case after hours.
- Action: It is likely the specimen was only recently changed to a rush (if it was originally a rush case, it would have been read earlier in the day). Ask the clinician if the patient is stable, and if clinical management would change overnight based on the results. In most situations, the patient will be stable--tell the clinician that you will notify the attending/resident via email, and they will follow-up the next morning.
- During the week: Rushes dropped off before 8pm should be grossed and loaded on the processor. If before 7pm, you can ask a PA or small gross tech to gross the rush.
- Rushes dropped off after 8pm should involve a conversation with the ordering/paging clinician regarding when the next day they need a read and whether the specimen is already in formalin. If they need it first thing in the morning and the specimen is already in formalin, then the on-call resident must go into the hospital to gross and load the specimen on the processor. Otherwise, it is okay to leave the specimen until the next morning, if it is already in formalin. Regardless, the RUSHblock should be emailed after the urgency is discussed with the clinician. Include in the email which clinician you spoke to, by what time they expected a read, and whether a PA needs to gross the specimen in the morning or if the specimen was already grossed and loaded onto the processor.
- For RUSH biopsy specimens received afterhours Sunday through Thursday that require fixation prior to being loaded on to a 4-hour processor, gross them in orange cassettes, email RUSHblock and the corresponding attending, and place the cassettes in the Tupperware labeled “2am processor, 4 hour run” located next to the 2am breast bucket. This Tupperware will be loaded by histology at 2am Mon-Fri morning.
- If you get a large specimen after hours that is a next day rush and has already been fixing in formalin, the specimen should be grossed. Send an email to the RUSHblock, including Jenn Patel, Sarah Ferguson, and Kara Tassinari, and they will ensure the rack gets loaded once a PA gets in the next morning. If the specimen is fresh/hasn’t been prepped and needs to fix for 8 hours, please forward this request to the RUSHblock, Jenn Patel, Sarah Ferguson, and Kara Tassinari, and it will get grossed first thing the next morning.
- Bone Marrow Biopsies that are labeled RUSH do not need to be processed by the on-call residents after the accessioners and PAs/small gross techs leave (~7 pm) during the weekdays, the bone marrow biopsies can be left in B+ until the following morning. Please contact the Hematopathology fellow and/or attending on service if there are any questions or if there is a RUSH bone marrow biopsy over the weekend.
- On the weekend: Any rush or clinically urgent cases should be accessioned, dictated, and grossed by the on-call resident. You must load all of the rush specimens unless they come in on Sunday after you have loaded the processors (unless a read first thing Monday morning is required by the clinician).
- Email the RUSHblock group email to alert histology about the rush case. Be sure to include the attending and resident that will receive the case.
- If you are notified of a rush being delivered after you have the loaded the processors on Saturday or Sunday, contact the clinician. If the clinician is insistent that the results of the slides will change clinical management of the patient and that this cannot wait until Monday, first talk to the on-call attending to make sure they are available for a Sunday read. If yes, you must gross the specimen, load it into the processor, and notify the RUSHblock..
- If it can wait until Monday morning, send an email to Jenn Patel, Sarah Ferguson, and Kara Tassinari asking them politely to gross the Sunday rush first thing when they get in on Monday morning.
- Always load processors after the specimen has fixed. Even if a processor is set to start later, specimens should be fully fixed before loading to ensure high quality tissue sections.
Transplant Cases
- Page/Question: Organ transplant people page you with a heads-up about a prospective transplant
- Action: Ask when the transplant is happening and ask where the team is now (Ask for the OR number, which will make it clear if they have an OR yet or not, and ask if donor is in OR). Tell them to page you again ~30min prior to the actual procedure. Notify them of our frozen policy between 8pm and 8am, that we will not be in house and need advance notice.
- Note: The transplant could happen at any time after they first notify you (or not during your shift, or not at all), so to minimize the waste of your time, ask for a more timely page (which may still involve a long wait, but it’s the best option). No matter when they tell you the transplant is happening, it will almost certainly be later than that. That said, if they say the frozen is in 1 hr and they have the OR, it’s best to stick around and be ready.
- When you cut the frozen, cut 2 H&E slides. The organ bank will bring a worksheet for the senior resident to fill out (re: histology of the organ). When they are done, they take one of the frozen slides with them, so in order for us to have an H&E on file, you should cut a second one at the time of the frozen.
- Evaluation and handling of liver allograft biopsies:
- Freeze the core entirely.
- Cut TWO slides for each specimen, as one slide will go to UNOS.
- The most important thing to evaluate is the percentage of macrosteatosis, as this correlates with post-transplant rejection (they like to see <30%).
- Complete the UNOS form, which includes additional details such as % macrosteatosis, % microsteatosis, extent of lobular/portal inflammation, fibrosis, and necrosis.
- Retain a copy of the UNOS form for MGH records.
- Label the UNOS slides with the patient’s name, UNOS number, and specimen type (e.g. right lobe of liver).
- Leave slides and paperwork at the frozen lab desk for proper accessioning in the morning (do not accession yourself). Leave a note/e-mail accessioning to inform them.
- Take the tissue down like a normal frozen and put in formalin.
- Evaluation and handling of renal allograft biopsies:
- Freeze the core entirely.
- Cut TWO slides for each specimen, as one slide will go to UNOS.
- The most important thing to evaluate is the percentage of glomerulosclerosis (i.e. the number of sclerosed glomeruli vs. total glomeruli).
- Note: They need 50 glomeruli evaluated at a minimum, so if you don’t count 50, get a deeper section and add the numbers together.
- Complete the UNOS form, which includes additional details such as extent of glomerulosclerosis, presence/extent of interstitial fibrosis, tubular atrophy, and arteriosclerosis/hyalinosis.
- Retain a copy of the UNOS form for MGH records.
- Label the UNOS slides with the patient’s name, UNOS number, and specimen type (e.g. right lobe of liver).
- Leave slides and paperwork at the frozen lab desk for proper accessioning in the morning (do not accession yourself). Leave a note/e-mail accessioning to inform them.
- Take the tissue down like a normal frozen and put in formalin.
Lymphoma Work-up
- In general, if there is sufficient tissue for multiple cassettes, perform a frozen section to triage the material. If you feel that the specimen is too small, page the HP fellow or attending on-call and ask how to handle the specimen.
- See this very helpful information sheet posted above the frozen lab bench.
- Often times, a specimen will come with a requisition that says “r/o lymphoma” but no callback is requested. In these cases, still follow the above guidelines and perform a frozen section to triage the tissue if enough material is available.
- Eye pathology samples (usually from MEEI) labeled "orbital mass" and received in the frozen section lab in saline should be sent for flow.
- Usually two parts are sent for "orbital mass" samples, a larger piece in formalin for permanent and a smaller piece in saline for flow cytometry; please confirm receipt of these two parts (by looking in EPIC or discussing with the surgeon/eye pathologist)
- The smaller piece received in saline can be sent directly for flow cytometry without performing a frozen section; discuss with eye pathologist on call if any questions arise
Flow Cytometry
- An eraser sized piece of tissue should be placed in Hanks and left in surg path with an accessioner to triage to Mayo via the Core Sendout lab. These should not be placed in the flow bin since they are no longer done on site.
Weekend rush leukemia/lymphoma flow at BWH
- On weekends, the Flow Lab Specimen pager will be forwarded to Hemepath pager. The Hemepath pager will be forwarded to the AP 1st call pager, 23305. Therefore, the AP on-call resident may initially field these calls and should involve the on-call HP fellow and attending as needed.
- MGH Hemepath on-call attending will determine priority for RUSH flow testing at BWH.
- Specimens received after 3 PM on Friday up until 11 AM on Saturday can be sent to BWH on Saturday for same-day processing.
- Specimens received after 11 AM on Saturday up until 11 AM on Sunday may be sent to BWH Flow Lab for same-day processing. If it won’t change clinical management to wait until Monday morning, then the sample can be held at MGH to be run as a RUSH on Monday morning.
- Specimens received after 11 AM on Sunday should remain at MGH to be run as a RUSH on Monday morning.
- Once the MGH Hemepath attending determines that RUSH flow testing at BWH is needed, someone from the on-call team needs to contact Core Lab at 617-726-3637 and request that a STAT courier be called to pick up the sample from MGH for delivery to the BWH * * Flow Cytometry Laboratory. If molecular/cytogenetics samples have also been received on the same patient, those should be sent via the same courier for delivery to BWH CAMD. Core Lab will separate and label the samples individually for delivery to the BWH Flow Lab and BWH CAMD. Sample must arrive at BWH Flow Lab by 2 PM for same day processing and results. Contact number for BWH Flow Cytometry Lab to confirm receipt: 617-732-5846.
- When sending a rush flow case over to BWH on a weekend (following discussion with and approval by the hemepath attending on-call), you should simultaneously send an email to BWHFlowCytometry bwhflowcytometry@partners.org and BWH LCO Client Services BWHLCOClientServices@partners.org.
- Cases will be accessioned in BWH PowerPath, will cross to Epic and should be viewable by both institutions.
- Cases will be interpreted and signed out by the BWH attending pathologist. Flow plots can be sent to MGH at a later date if they are needed for MRD reference.
- HOLIDAYS other than Sat/Sun: BWH does not staff flow cytometry on holidays, but they try to have someone trained in flow in the main hematology lab on holidays in case of emergencies. If there is no flow person available for the holiday, one of the flow techs will be on call. They do not carry a pager. Instead, call the main hematology lab (617-732-6850) to coordinate the RUSH testing.
- For organ transplant T cell monitoring- redirect questions to Dr. Preffer (p23319)
Products of Conception
- Look up the patient history. Often times, the specimen is for “r/o ectopic”. Notify the senior resident. Float the tissue in a saline-filled Petri dish to identify villi (dissecting microscope in the frozen section lab can be helpful).
- If villi are definitively grossly identified, notify the clinician and submit one representative section of the villi.
- If no villi are definitively grossly identified, notify the clinician and submit the entire specimen as a RUSH for permanent evaluation.
- For other types of POCs, contact the clinician to ask what their question is, how urgently the specimen needs to be processed (i.e. over the weekend or Monday morning), and whether additional studies need to be sent (e.g. cytogenetics). You can also loop in the OB attending on-call afterwards via email.
- If you have any request for karyotype (cytogenetics) on a POC or fetal tissue please follow the steps outlined below. 1) Use sterile kit to identify villous or fetal tissue. 2) Put villous/fetal tissue into cytogenetics media tube. 3) Label with Copath label. 4) Print cytogenetics order requisition from EPIC. 5) Write FOR QUEST in bold lettering at the top. 6) Write the sample into the send out logbook 7) Send the sample and cytogenetics requisition to CORE LAB clearly labeled for QUEST.
Prepping Specimens
- If the Junior hasn’t prepped a specimen before, he/she should prep them under direct supervision of the Senior. There are detailed descriptions on how to prep all specimens in the grossing manuals on the Hub; however, it is still best for the Senior to support the Junior and be present with them at the bench when they are learning how to prep. “’Don’t forget to put formalin stickers on all prepped specimens!!”’
- Most small specimens: are already in formalin, just put them on the shelf as they are.
- Most medium sized specimens: gets tossed into formalin except...
- Fat pad biopsies: for amyloid (half frozen for IF and half into formalin for permanents).
- Gallbladders: open before tossing into formalin, be sure to put stones in the jar so grossing staff knows they were there.
- Bowels: Open them before putting them in formalin! Open them before putting them in formalin! Open the ileocecal valve in right hemi-colectomies! Pin out if removed for tumor or suspicious for tumor. Sometimes figuring this out is harder than you think! The pre-existing diagnosis is not infrequently wrong, so use your judgement.
- Heart explants: weigh, photograph front and back, freeze a piece of left ventricle for IF, and put a tiny piece into EM fixative. Put in formalin.
- Liver explants: weigh, photograph, take the margins (hepatic veins and hilar margins = bile duct, hepatic artery, and portal vein), slice at 0.5 cm intervals, stuff with paper towels to ensure fixation. Put in formalin.
- Lung explants: weigh, photograph and take bronchial and vascular margins. Put in formalin.
- Breast specimens: Do not need to be grossed unless explicitly asked by the breast attending. Breast specimens that are held into the weekend or overnight have been prepped and placed in formalin for the per diem PA to gross or will be grossed the following day by PA staff. Mastectomies: weigh, ink, section through, put in formalin. Reductions: weigh, section through, put in formalin.
- Uteri: Weigh, ink (if appropriate), bivalve and photograph. No need to slice up as you would on frozens, leave it otherwise intact. Put in formalin.
- Kidney: Check if it’s a medical renal or tumor case. If tumor: weigh, ink, take margins and bivalve, then put in formalin. If medical: check the renal path grossing manual .
- Fragmented Leiomyoma: Weigh and add formalin.
Uncommon Biopsies
Fat pad biopsies
- Cut it in half, save half for frozen, and put the other half in formalin.
- Page/Question: How big should a fat pad biopsy for amyloid be?
- Answer: Per Dr. Stone, it should be 1.0 cm^3 in size.
Cardiac biopsies
- Usually received on ice. Do NOT use a chuck to freeze it. Write the patient information (Name, MRN, Accession Number) on the back of a biopsy card. Freeze by putting a drop of OCT on a biopsy card or directly into the cryostat and embed the cardiac tissue within it. After the drop freezes, attach the CoPath container label to the paper (see below), and put it into a small bag with a copy of the requisition form. Scan the Asset label to “SPUF” (SPU Freezer for Frozen Saved Tissue) and put the asset label in the bag. Place it in the -80C fridge for Warren 5.
- Apical core biopsy (received fresh, not on ice):
- Accession the part type as "Heart Exc Left Ventricle" (so it'll be properly processed; using “heart biopsy” generates a different protocol)
- Save a piece frozen (don't forget the asset label; see below)
- Save a piece for EM.
- FYI: Using a chuck creates a plane between the chuck OCT and the OCT added on top, which creates processing issues.
- Note: There should be another portion of the specimen for permanents (which is usually accessioned and handled by the accessioners during the day). After hours and over the weekend, if you cannot find another portion for permanents, check with the clinical team before freezing the tissue.
- After hours or weekend/holiday endomyocardial biopsies for transplant rejection: Per JST, the on call resident should come in after hours to freeze the piece for IF. Rush endomyocardial biopsies accessioned over the weekend are for a Monday read (or next business morning if a holiday weekend).
Muscle biopsies
- As soon as you hear that a muscle biopsy is coming, contact the SPU manager (if during regular hours)
- If after hours, use the on-call tech list (posted on the inside of the SPU door on Blake 3) to call the first on-call tech and ask if someone from SPU will be in/can come in to process the muscle.
- If no SPU staff are available, you will process the muscle.
- Call your senior, use Javier’s instructional video, or reference the 10-step guide to Muscle Biopsy on MediaLab for help.
- Remember to save a piece of muscle for electron microscopy and to enter a gross in CoPath: "Received labeled, measurements, portion sent for EM and remaining to SPU"
- Once complete, send an email to SPU staff at MGHIHC-SPUStaff@partnershealthcare.onmicrosoft.com
- If this happens, notify the chief so that we can keep track of how often this is occurring.
Temporal artery biopsies
- Any temporal artery specimen (regardless of whether or not marked RUSH) should be treated as a RUSH and grossed/processed right away.
- Do not cut them. Wrap each temporal artery in histowrap, place in a orange cassette with a ribbon, and write “temporal artery” in pencil on the side of the cassette.
- If a frozen section is requested to confirm if artery is present, shave the two ends of the fragment and freeze the cross-sections. Temporal arteries from Mass Eye and Ear accessioned to Eye Path as Rushes on Fridays, in recent experience, have been treated as Monday rushes, per Eye Path request (ie, they are not re-accessioned to CV for Saturday read). Always a good idea to check in with Eye Path just to confirm though.
Skin biopsies for immunofluorescence
- Biopsies are usually delivered to Pathology in Michel’s transport medium to be processed for immunofluorescence studies. The tissue can be left in Michel’s medium for up to 72 hours (preserves tissue and its antigenicity), so for weekdays and normal weekends, your only job is to make sure the sample is placed in Michel’s medium (do NOT leave it fresh or try to freeze it yourself). The IF lab will process it the next business day.
- If a clinician calls you looking for Michel’s medium and they are unable to obtain it on the clinical side, we have a small stock either in Blake 354 by the accessioner’s window. The medium can be used even if there is salt precipitation around the lid.
- Dermatology is responsible for sending their specimens in Michel’s medium--if they ask you for it, let them know that is their responsibility for future reference (but of course provide it to preserve the current specimen).
- Exception: The ONLY time you should freeze the biopsy yourself is if it’s the Friday afternoon/evening of a holiday/long weekend and it’s possible the tissue will sit in Michel’s medium for longer than 72 hours before it can be frozen by the IF lab. Freeze it in OCT on a chuck (following the same procedure as cardiac biopsies above) and store it in the -80 C freezer for Warren 5.
- Another situation is If you and the clinician are unable to find Michel’s medium. Don’t panic. Michel’s medium is a transport medium, not a fixative. It preserves tissue for IF for up to 5 days but does not actually do anything to “fix” it. In this case, ask that they personally and expeditiously bring the specimen to you in the frozen lab in saline and freeze it once they deliver it to you.
- Check with someone more experience if you are unsure about how to orient the specimen for freezing.
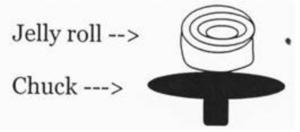
Skin “jelly roll”
- At night or on the weekend, you may rarely get a skin “jelly roll” with the clinical history of “r/o SJS vs. TEN.” Basically this is a rolled up portion of sloughed skin that should be cut like this in order to evaluate the extent of involvement of the epidermis:
Rush kidney biopsy for transplant rejection
- Rush kidney biopsy for transplant rejection:
During work hours, IR-guided kidney biopsies for transplant rejection are checked by a histo tech for adequate glomeruli at the time of biopsy. After hours and on weekends/holidays, the senior resident on call should assess these biopsies for adequacy (at least 7-10 glomeruli seen using the dissecting microscope).
Cytogenetics
- Weekdays:
- Last pick-up around 5:30 pm; prior to this time, give to one of the accessioners (labeled tube, completed cytogenetics form, and photocopy of requisition sheet.)
- After 5:30pm, the specimen can be left in the fridge overnight (prepared as above), and the accessioners will take care of it in the morning (leave a note to let them know it is there!).
- Weekend cytogenetics policy:
- Special courier transport of cytogenetics specimens to BWH CAMD over the weekend should only be performed for the following clinical circumstances: 1) r/o acute promyelocytic leukemia (APML or APL) or 2) r/o Burkitt lymphoma. If the history is not clear based on the requisition and chart, please page the clinician, HP fellow or attending on-call for clarification.
- RUSH cytogenetics for heme malignancies is typically sent from a bone marrow or peripheral blood specimen, but the workflow is currently being expanded to include other types of specimens (including other fluids).
- The core lab typically transports cytogenetics specimens to CAMD. However, if they cannot do it, you must arrange a courier to transport the specimen (see below).
- The cytogenetics fellow on call should be contacted regarding any case that is sent to BWH CAMD over the weekend (pager number 30527). Please also alert the CAMD lab to the RUSH status of the specimen by emailing BWHCAMDAccessioning@partners.org.
- All other specimens are to be kept at MGH for delivery to BWH CAMD on Monday morning. If it is a 3-day weekend, the specimens will be delivered on Tuesday. Specimens should be kept in the refrigerator over the weekend.
The cytogenetics fellow on call should be contacted regarding any case that is sent to BWH CAMD over the weekend (pager number 30527). Please also alert the CAMD lab to the RUSH status of the specimen by emailing BWHCAMDAccessioning@partners.org.
All other specimens are to be kept at MGH for delivery to BWH CAMD on Monday morning. If it is a 3-day weekend, the specimens will be delivered on Tuesday. Specimens should be kept in the refrigerator over the weekend.
Courier Instructions
- Current instructions are posted on the bulletin board above the middle accessioning station and a photo is attached
- Call Skycom courier as soon as you are aware that they will be needed
- Phone: 617-782-1130
- Account: A2217
- Cost Center: 1200-952600-MG-7524
- Destination address: 75 Francis St, Boston MA; Shapiro Bldg 5th floor rm 032
- After calling the courier page cytogenetics and call them at 1-857-307-1500
- CAMD is in house Saturdays and holidays 8am-4pm.
Autopsy
- Occasionally, we are paged to answer questions about getting autopsy permission. You should be able to answer most of the questions concerning obtaining permission after a week on the autopsy service. If there are any questions you cannot answer, contact the senior resident on-call or the autopsy attending on-call. The inpatient floors should all have copies of the autopsy permission form.
- If you are notified about an autopsy that is coming, email the details to the autopsy attending, the Autopsy PA Group, Autopsy Staff Group, and the resident on autopsy who will be getting the case.
- Occasionally, we are paged to answer questions regarding body transport and delivery. It is the responsibility of whoever is transporting the body to us to arrange body transport via a funeral home. If there are questions regarding directions to the hospital, where to drop off the body, or needing access to the morgue, the person can be directed directly to MGH security (617-726-2121).
- If New England Organ Bank (NEOB) pages you overnight to request use of the morgue, tell them that is okay and direct them to security at (617) 726-2121 to open the door. You should also notify the Autopsy team. If the autopsy is pending an MGH autopsy (check admitting ext 63384) and guidance is needed about possible restrictions on specific organs, call Justin Susterich.
- We no longer perform weekend clinical autopsies effective April 1 2023. Per Justin, please note that pages/communication about outside cases coming to MGH over the weekend from affiliate hospitals will be directed to the on-call resident same as weekday after hours. These hospitals do their own consent and will send the decedent over automatically. All the on-call resident needs to do is acquire the clinical information and questions for autopsy from the paging pathologist/clinician and email Monday’s autopsy team the information. If we receive a call about requesting an outside autopsy from a family, then the decedent affairs/techs would get that call and coordinate an email. Claudia and Ariana will still be here on the weekends grossing/accessioning and can help residents if they have any questions about autopsy-related pages that they receive. You should not receive a request for performing a weekend consent with this new system, but if you do, you can defer to Monday’s team.
Call Room Resources
There are both a Male and Female call room at MGH. The Female Call room is located on Bigelow 9 (952T) all female trainees should have access to this room. The Male call room has been temporarily relocated from Wang 125 to Austin(Founders) 8th Floor. There are signs located outside of the space when you get off the elevator. All male trainees should have access to this space.
Tissue for Research
- If you receive a request to bank tissue for research after hours or on the weekend, please politely explain to the person requesting that we are only able to bank during the hours of 7AM-6:30PM Monday to Friday and under the supervision of a PA and/or attending pathologist. No banking of research specimens on the weekend unless a PA happens to be present or collaborating pathologist is available (in either case, “’RESIDENT DOES NOT BANK ON WEEKEND”’). Residents can also reassure the requestor that tissue will remain fresh (i.e. not placed in formalin) in the fridge for supervised collection to occur the morning of the following business day and offer them that option. If you get pushback, feel free to tell them they can contact Dr. Aliyah Sohani as Director of the Lab to discuss further.
- The on-call team will not bank or provide tissue to research teams after hours Monday to Friday or over the weekend.
- Please refer any onerous requests to the program directors and Aliyah Sohani.
Neuro Tissue for Research
- After 5pm, the senior on call is responsible for releasing neurologic tissue for research requests. All questions should be directed to NP staff and not to fellows.
- First, before any tissue is released, there must be a frozen section diagnosis obtained for the specimen. If none has been performed then a portion of the tissue being submitted for permanent histology should be frozen to confirm the diagnosis.
- For all cases, the senior resident should assess if the amount of tissue being submitted for permanents and research is a reasonable amount. In general, we always want to keep more tissue for permanents than is given away for research. If it seems unreasonable then I have taken some tissue out of the research container and added it to the permanent container (trying to be as sterile as possible if sterility is needed for research). I feel most comfortable when we are giving away only 10-20% of the tissue, though more can be ok if there is a lot in both containers and we will have plenty to make a diagnosis.
- If those conditions are met, cases that can be released by the senior resident without consulting the neuropathology attending on service that day include:
- Schwannoma
- Pituitary adenoma
- Meningioma
- Glioblastoma or a Grade IV astrocytoma that has definitive high grade features on frozen (necrosis and/or microvascular proliferation)
- Recurrent/residual glioma that shows grade 4 features (necrosis and/or microvascular proliferation)
- Metastases
- Cases that should always be discussed with the neuropathology attending who saw the case in the frozen lab include:
- Low-grade gliomas
- High-grade gliomas that don’t have features to warrant being called GBM or grade 4 on frozen
- Anything else not listed here
- The general principal is we don’t want to give away too much tissue on a case that the grade could change if we see certain diagnostic features by examining more tissue.
- Also, if there is no researcher present to collect the tissue, please make every attempt to contact them and let them know it is available to pick up. Leave the research portion in the fridge next to accessioning and make sure the PAs know to look if the specimen is still there in the morning. If it is not picked up by the morning then the PAs should add it to the permanent tissue submitted. Please send an email to the neuro fellow that a piece was left in the fridge for a researcher to pick. NO page necessary.
Cytology After-Hours Policy
- Cytotechnologists are available for rapid onsite evaluation (ROSE) of cytology smears that are ready and available for review until 5pm Monday-Friday
- If a rapid is requested after 5PM, the clinical team should bring the smeared slide to the frozen section lab and the on-call team should stain the slide following the typical H&E procedure in the frozen lab and then contact the on-call cytology attending, who will either come to the frozen lab to read the slide or may request to view the slide over Teams
- On-call residents are not responsible for picking up the specimen from the OR, for smearing the slide or for reviewing the cytology slide
- Contact the cytology B on-call cytology attending from Monday to Thursday and the cytology attending on call for the weekend from Friday to Sunday
- Cytopathology specimens are not processed on the weekends or holidays. After hours, all specimens are delivered to the core lab receiving area where transport will deliver the next business day or cytopathology will go up and retrieve them early in the morning of that business day.
- If you receive a rush cytology request after hours/weekend/holidays: email Ron Arpin and Nathalie Kumiega; they will process the request on the next business day.
Swapping Call Days
To swap call days, make sure to first confirm the swap with another junior resident, then ask a Chief Resident to change both residents’ initials on the Junior Call Schedule, which will get pushed to LearnPath.